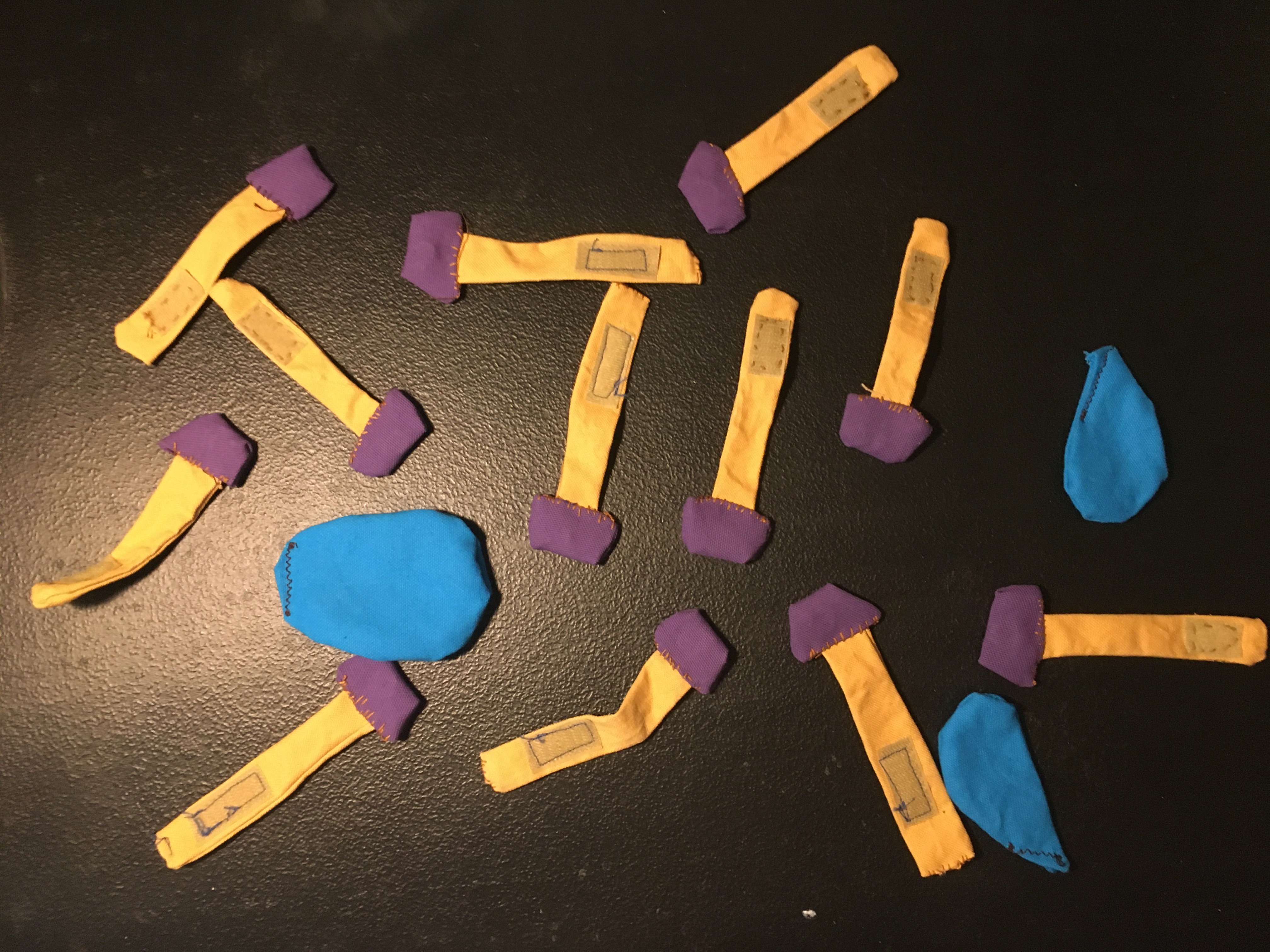Back in 2018, I had the opportunity to participate in the Pacific Science Center’s Science Communication Fellowship Program. Not only did I take classes on science communication, I made my very own, hands-on demo of my research topic.
My graduate research focused on how water interacts with soil, addressing the question of why soil sometimes does not absorb water from a molecular standpoint. Most rocks and minerals are readily wettable, so any water resistance must come from the other major component of soil: the organic material from soil microorganisms and decaying plants. I focused on one particular class of molecule, the phospholipid, which forms cell membranes. One end of a phospholipid readily interacts with water; the other does not. The hydrophobic ends of molecules like these were hypothesized to cause soils to repel water. I wanted to find out if and how that might occur.
Demonstrating the whole scale of my project was a big hurdle for designing my demo. I opted to start at the macroscopic and work my way down to the molecular.

Starting off with a jar of dirt, I asked participants to tell me what they saw inside the soil. Typically, participants were quick to find small rocks. I would then ask them if they saw the twigs and pine needles as well. This way, I introduced the concept of soil’s two major components: inorganic (rock and mineral) and organic (plant and animal matter).
Next, I had a box full of plush mineral grains, single-cell organisms, and plant detritus. I asked participants if they could find all the soil components they saw in the real dirt in the box. I included a big drop of water to spark conversation about how water might move through soil. Most participants guessed quickly that the purple blobs were bacteria and other microorganisms, which let me segue into talking about cell membranes and the molecules that make them up.

Lipids can self-aggregate in a number of ways depending on their surroundings; they have polar ends that interact with water, and long carbon tails that do not interact with water. To demonstrate the behavior of lipids, I made model lipids with magnetic “heads” and velcro “tails.” I also made magnetic water droplets to demonstrate how the different ends of lipids behave. Once visitors had played with the molecules and figured out how they interacted with each other and water, I would ask them to form their own hypothesis for how lipids could make soil hydrophobic. (See photo gallery below. Purple = lipid head groups, yellow = lipid tails.)
Having now done this demo several times, I’ve seen that the magnetic/velcro molecules get the best reception from visitors. Once in-person interaction resumes, I plan to revamp the activity to mostly focus on the molecular scale. I will make magnetic “mineral grains” so that participants can build the lipid structures that have been hypothesized to form on mineral surfaces. As a bonus, since I’ve now finished my Ph.D. project, I can tell them if their results match mine!